Middle school science teachers! Are you teaching acids and bases? You’ve come to the right place! I know it can be a tricky topic to teach. It’s abstract, and it’s easy for the kids to get confused, especially when it comes to pH. Here’s how I teach it.

Prior Knowledge
First off, I ask them what they already know. What acids or bases have they encountered in their everyday life? You’d be surprised by some of their answers! Some of them will know that a lemon is acidic, or that orange juice is acidic (they usually know far more about acids than they do about bases). Some of them will guess Coca-Cola is acidic, maybe even a car battery. I’ll bet some of them will know about acid rain. And you know that at least one of them will ask what the strongest acid in the world is (they’re fascinated by this, it’s fluoroantimonic acid, here’s a video showing what it can do.)
pH Levels / pH Scale
In a general science classroom, I don’t go into too much detail on the strict definition of pH. It’s far too advanced for them. I just say that it’s how we measure how acidic or basic something is, and then I dive straight into showing them the pH scale. I don’t mention hydrogen ions, hydroxide ions or any of the advanced stuff. That’s for an advanced physical science/chemistry class later in high school. The pH scale is the basic building block in teaching acids and bases. It shows whether something is an acid, neutral, or base. It also shows how strongly acidic or basic a substance is. I teach them that the further away they go from neutral (pH 7), the stronger the acid or base is. Weak acids and weak bases are close to pH 7. Strong acids and strong bases are further away.
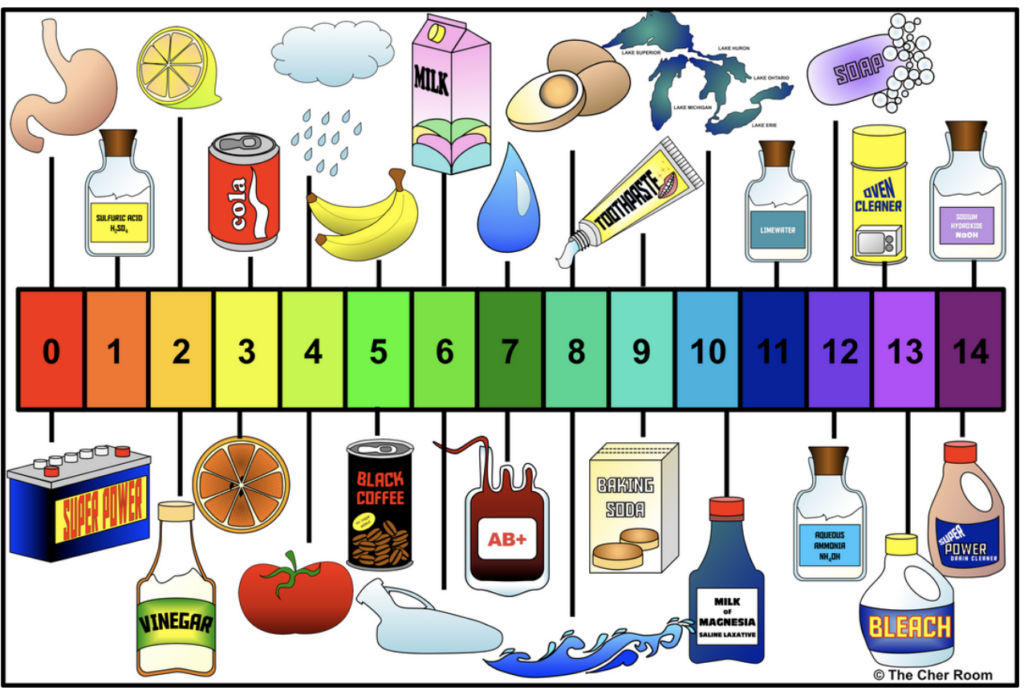
pH of Soils
This is a great time to talk to them about the different pH of soils. Again, some of them may already know this. Some plants need to grow in acidic soils like blueberries and hydrangeas (more on these in a sec!). Others prefer an alkaline soil, such as lavender or asparagus. Some prefer a neutral (or very slightly acidic) soil, such as tomatoes, peppers, and roses. The flowers of hydrangeas change color depending on the soil they’re in! In acidic soil, they’re blue; in alkaline soil, they’re red. So, hydrangeas are a natural indicator of soil pH that they see in their daily lives! Quick aside: I hate hydrangeas. I know, how can you hate a flower? I mentioned this in class one day when I was teaching acids and bases, and a little girl at the back looked shocked! They were her favorite flower!
Properties of Acids
Next, I ask them about common examples of acids they already know, e.g., lemon juice and Coca-Cola. Is there anything common to these substances? They’ll usually mention that they are bitter/ sharp. I mention stomach acid because it has such a low pH. This is usually a wow moment for them! You can also tell them about formic acid, which ants use in their sting.
Properties of Bases
This is more difficult for them, so I mention that most bases are cleaners, e.g., toothpaste, oven cleaner, drain cleaner, laundry detergent, etc. I talk about the common lab bases such as sodium hydroxide (and where it’s found in daily life).
Next, we get into the basic chemistry and indicators. How can we tell if something is an acid, has a neutral pH, or is basic? And going further, how can we see if one acid is stronger than another or if one base is stronger than another? I talk about common indicators, such as the lab favorite red cabbage juice! Here’s a simple recipe to make it:
Red Cabbage Indicator

1. Chop a cup of red cabbage leaves.
2. Add about half a cup of water.
3. Blitz it in a food blender (NutriBullet, etc) until it becomes a liquid.
You can strain it through a cloth if you like but it isn’t necessary. Red cabbage juice is safe, cheap to make, and lasts for a few days (3-5) in the fridge. It’s perfect as a DIY indicator.
Here’s a color guide:
| pH Level | Color | Example |
|---|---|---|
| Very acidic | Red | Lemon juice, vinegar |
| Acidic | Purple-pink | Soda |
| Neutral | Purple | Water |
| Basic | Blue-green | Baking soda solution |
| Very basic | Green-yellow | Liquid soap, bleach |
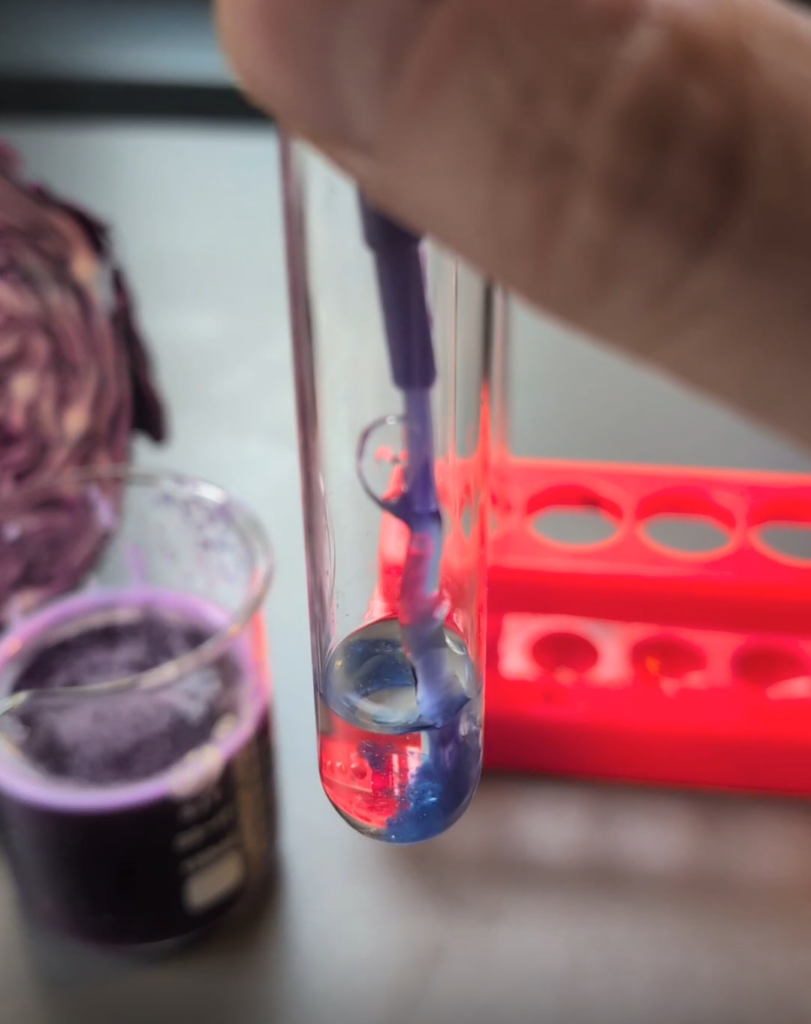
It works great! If you don’t have litmus paper, red cabbage juice is a great alternative. Just add a few drops of the cabbage juice indicator to the test tubes to see the color changes. It makes this a fun, interactive lesson that kids remember.
Test it on these common household items:
- Vinegar
- Lemon juice
- Soda
- Milk
- Water
- Salty water
- Baking soda water
- Soap
pH Scale Worksheets
Here’s a link to a free pH scale worksheets activity. It’s a 4-page worksheet which also includes a student worksheet and a pH indicator of common items handout. Both these are printed two/page for easy distribution. The aim of the worksheet is to familiarize students with the pH scale and to show where acids and bases are located. It will test students’ knowledge on the pH scale.
For chemical reactions and neutralization reactions, a simple experiment is to add hydrochloric acid to sodium hydroxide. Be very careful using these chemicals in your school laboratory, as they can be very corrosive. Dilute them to reduce the chances of an accident. This can be a fun lab. You can test if neutralization has happened using litmus paper, red cabbage indicator, or a pH meter. You can let the solution evaporate over a few days and students will be able to see the salt (NaCl) left behind. Here are lesson plans on acids and bases from my TPT page. It also includes an acids and bases PowerPoint.
pH Scale Doodle Notes Activity
Students love doodle notes! You can use this pH scale worksheet to let students doodle and color in the worksheet.
Make sure you download your acids and bases worksheet here. Middle school students love it!
